PROOF OF CONCEPT/PILOTS/METHODOLOGIES
Securing the Chain of Custody and Integrity of Data in a Global North–South Partnership to Monitor the Quality of Essential Medicines
Kathleen Hayes1  ; Natalie Meyers, MA, MLIS2
; Natalie Meyers, MA, MLIS2  ; Christopher Sweet, PhD3
; Christopher Sweet, PhD3  ; Ayenew Ashenef, MS4
; Ayenew Ashenef, MS4  ; Tim Johann, PhD5
; Tim Johann, PhD5  ; Marya Lieberman, PhD1
; Marya Lieberman, PhD1  ; and David Kochalko, MBA, MA, MPP6
; and David Kochalko, MBA, MA, MPP6 
1Department of Chemistry and Biochemistry, University of Notre Dame, Indiana, USA; 2Lucy Family Institute for Data & Society, University of Notre Dame, Indiana, USA; 3Center for Research Computing, University of Notre Dame, Indiana, USA; 4Department of Pharmaceutical Chemistry, School of Pharmacy, College of Health Sciences, Addis Ababa University, Ethiopia; 5Department of Chemistry, Roanoke College, Salem, Virginia, USA; 6ARTiFACTS, Cambridge, Massachusetts, USA
Keywords: analytical chemistry, blockchain, ledger, pharmaceutical, quality of medicine, supply chain
Abstract
Substandard and falsified (SF) pharmaceuticals account for an estimated 10% of the pharmaceutical supply chain in low- and middle-income countries (LMICs), where a lack of regulatory and laboratory resources limits the ability to conduct effective post-market surveillance and allows SF products to penetrate the supply chain. The Distributed Pharmaceutical Analysis Laboratory (DPAL) was established in 2014 to expand testing of pharmaceutical dosage forms sourced from LMICs; DPAL is an alliance of academic institutions throughout the United States and abroad that provides high-quality, validated chemical analysis of pharmaceutical dosage forms sourced from partners in LMICs. Results from analysis are reported to relevant regulatory agencies and are used to inform purchasing decisions made by in-country stakeholders. As the DPAL program has expanded to testing more than 1,000 pharmaceutical dosage forms annually, challenges have surfaced regarding data management and sample tracking. Here, we describe a pilot project between DPAL and ARTiFACTs that applies the blockchain to organize and manage key data generated during the DPAL workflow, including a sample’s progress through the workflow, its physical location, provenance of metadata, and lab reputability. Recording time and date stamps with these data will create a permanent and verifiable chain of custody for samples. This secure, distributed ledger will be linked to an easy-to-use dashboard, allowing stakeholders to view results and experimental details for each sample in real time and verify the integrity of DPAL analysis data. Introducing this blockchain-based system as a pilot will allow us to test the technology with real users analyzing real samples. Feedback from users will be recorded and necessary adjustments will be made to the system before the implementation of blockchain across all DPAL sites. Anticipated benefits of implementing the blockchain technology for managing DPAL data include efficient management for routing work, increasing throughput, creating a chain of custody for samples and their data in alignment with the distributed nature of DPAL, and using the analysis results to detect patterns of quality within and across brands of products and develop enhanced sampling techniques and best practices.
Citation: Blockchain in Healthcare Today 2022, 5: 230 - http://dx.doi.org/10.30953/bhty.v5.230
Copyright: © 2022 The Authors. This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0.
Received: December 16, 2021; Revised: December 21, 2021; Accepted: February 21, 2022; Published: March 21, 2022
Competing interests and funding: One of the authors is employed by a for-profit entity, ARTiFACTS.ai (Artifacts of Research, Inc.), that provides the platform utilized in the pilot described in this article. ARTiFACTS provides a complementary service for the BHTY journal, which allows the authors published in BHTY to link supporting evidence with their manuscripts and receive citations to that evidence. ARTiFACTS intends to apply the learnings from this pilot to determine the viability of developing a commercial solution.
This research work was funded by the following sources:
- Ethiopia Ministry of Innovation and Technology (MInT, https://mint.gov.et/) grant “Developing, validating and adopting simple mobile technologies in drug quality evaluation and counterfeit detection”
- NSF CMMI 1842369 “EAGER: ISN. Unraveling illicit supply chains with a citizen science approach” (https://www.nsf.gov/awardsearch/showAward?AWD_ID=1842369)
- Walther Cancer Foundation Grant #0178.01 “Developing Paper Analytical Device for detection of counterfeit chemotherapy (Chemo-PAD) in Kenya” https://www.walther.org/grants/
- Pfizer Foundation, Inc. (EIndiana: 13-6083839) Antimicrobial Resistance Project Moi Teaching and Referral Hospital Grant 419 (GR000419). “Implementing a model of improved care for infectious diseases and antibiotic stewardship across multiple levels of the health system in Western Kenya” https://www.pfizer.com/purpose/responsibility/healthcare-access/global-health-grants
- 2019 Blockchain Initiative award (Internal UND funding—https://research.nd.edu/news/notre-dame-blockchain-initiative-announces-seed-grant-recipients/)
- Roanoke College and Roanoke College Department of Chemistry.
Corresponding author: Kathleen Hayes, Email: Khayes5@nd.edu
An estimated 10% of pharmaceutical products in low- and middle-income countries (LMICs) are substandard or falsified (SF). Substandard drugs are authorized medical products that do not meet their quality standards and/or specifications, while falsified drugs are medical products that are deliberately misrepresented in their identity, composition, or source.1 SF drugs are responsible for adverse human health outcomes including failure to treat and prevent illness, leading to increased morbidity and mortality, as well as decreased trust in medical care systems.1,2
One of the reasons that SF drugs are so prevalent in some LMICs is a lack of widespread, post-market and pre-market quality testing of pharmaceutical dosage forms.3 As of 2017, 26 of the 54 drug regulatory agencies in Africa did not have a pharmaceutical quality control lab, and 40 did not conduct regular post-market surveillance activities.4 Most pharmacopeia assays for pharmaceutical testing rely on high-performance liquid chromatography (HPLC), which can be used to measure the amount of the active pharmaceutical ingredient (API) in a given dosage form – a key indicator of medicine quality. However, HPLC is a scarce resource in LMICs, as it is prohibitively expensive, requiring highly trained personnel, high-cost instrumentation, reagents, and consumables, as well as resources for maintenance and repairs.
The Distributed Pharmaceutical Analysis Lab (DPAL) was established in 2014 to leverage the testing capacity of colleges and universities in the United States and abroad to assess the quality of drugs that are collected in LMICs.5 Most colleges and universities have the capability to carry out HPLC analysis, which is the gold standard for pharmaceutical analysis. Once participants have carried out a series of careful experiments to demonstrate system suitability – showing that their HPLC system works for the analysis of a desired pharmaceutical product– that DPAL site can measure the amount of API in a given dosage form and compare it with the accepted amount of API required to qualify a dosage form as good quality. While this API content assay is only one component of quality testing, it is a key indicator of quality problems that can cause adverse patient outcomes. DPAL allows more dosage forms collected in LMICs to be tested for quality than would otherwise be analyzed through post-market surveillance.
Dosage forms undergo two types of sample analysis throughout the DPAL workflow. In addition to the HPLC-based sample analysis of DPAL, dosage forms are screened in-country at collection sites using a paper analytical device (PAD: a cost-effective tool for field screening of pharmaceutical dosage forms in low-resource settings.) The PAD is a field-friendly device that is used to screen drug dosage forms to identify suspicious products, prior to HPLC analysis.6 Samples that are flagged as suspicious via PAD screening can then be prioritized for HPLC analysis.
Throughout the past 7 years that DPAL has existed, over 1,000 dosage forms have been analyzed and 168 SF drugs have been detected through the program, including falsified acetaminophen, adulterated amoxicillin-clavulanate and doxycycline, and substandard losartan.5 This number of detected SF drugs is in alignment with estimates that 10% of drugs in LMICs are SF. The analysis results collected through DPAL are communicated to the stakeholders including the pertinent regulatory agencies and partner organizations in the country where the samples were collected and can be used to inform purchasing options.
Throughout the workflow of DPAL, a large amount of data are generated, stored, and shared; the physical location of each sample, its progression through the analysis workflow, results of the analysis, system suitability information, requests for samples, and alerts that samples are on their way to a location. All these data are currently housed on a myriad of platforms, including Open Science Framework (OSF), Excel, Dropbox (a file-hosting service), email, external hard drives, and personal computers. In addition, changes are made to existing data by several users, with no way to log or track the changes. The lack of streamlined data management threatens the security of the data, which means that the DPAL program does not operate as efficiently as possible. To mitigate the challenges associated with the data management in DPAL, the ARTiFACTS team is developing a blockchain database for managing DPAL data, which allows the system to be secured and show provenance of data. Founded by a team experienced delivering solutions widely used for scientific research, ARTiFACTS had blended the creation of cloud-based information platforms tethered to trusted distributed ledger technology. Employing Web 3 technologies, ARTiFACTS offers provenance security services for researchers across all disciplines and partners with universities, publishers, and other organizations seeking solutions for providing timely and verifiable access to information with scientific, societal, and commercial value.7–9
Blockchain has been adapted for numerous applications in science and health care that include managing research evidence, collection of informed consent for clinical trials, and confirming patient identity, with benefits for building a secure platform for managing and storing data.10–12 Incorporating blockchain in the technology stack for these and other applications introduces an enhanced layer of trust and verifiability, both of which are essential with use cases involving multiple participant contributors engaged in geographically distributed processes. The key benefits of integrating blockchain into healthcare applications include decentralized management, an immutable record of actions and data, robustness and availability, and enhanced security and privacy.13,14
Applying blockchain to mitigate DPAL’s current shortcomings is expected to enable the testing and sample handling workflow to operate more efficiently. Actions performed are recorded locally but are not visible to other participants. Controlled handling of samples and the provenance of results obtained cannot be verified. This approach is unable to scale and process the volume of samples required to maintain active monitoring and reporting of drug quality. The use of an interactive platform by all contributors, operating 24/7 that records all actions and scientific metadata to a blockchain ledger, is expected to speed handling, reporting, verification and management activities among multiple parties working from multiple geographic and organizational locations. Benefits include streamlining all data, results, requests, and physical sample location information into one platform, having record of provenance so we know when and by whom changes were made to data, and ensuring that updates can only be made by authorized personnel.
We describe, here, plans for a blockchain system to manage DPAL data, which uses a subset of DPAL participants as a model for the larger system. The blockchain-based system is being designed by an ARTiFACTS team. The DPAL participants included in the pilot are the in-country coordinator from Addis Ababa University (Addis Ababa, Ethiopia), the DPAL manager from the University of Notre Dame (ND) (Notre Dame, Indiana, USA), and the DPAL site coordinator and analyst(s) at Roanoke College (Salem, Virginia, USA). After this pilot, the use of blockchain will be expanded to all DPAL participants, including other collaborators in LMICs and other colleges in the US and abroad.
DPAL
The Distributed Pharmaceutical Analysis Lab leverages the testing capacity of academic institutions in the US and abroad, including LMIC institutions to test drugs sourced from LMICs. The major players in the DPAL workflow include the coordinators at each collection site, the DPAL manager at ND, the site coordinator and analysts at each DPAL site, and stakeholders, such as pharmaceutical regulatory agencies.
Workflow
The process of DPAL occurs in multiple countries and multiple locations, with each location contributing different data to the overall database, as outlined in Figure 1.
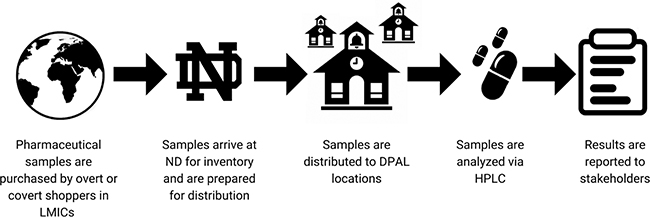
Fig. 1. The DPAL (Distributed Pharmaceutical Analysis Lab) process occurs in multiple countries and locations. Each location contributes different data to the overall database, as illustrated here. HPLC: high-performance liquid chromatography; LMICs: low-and middle-income countries; ND: University of Notre Dame.
- Collection: The DPAL process begins in-country, where overt or covert shoppers visit local pharmacies and other places that sell medicines to purchase finished pharmaceutical dosage forms of drugs, such as amoxicillin, azithromycin, and ceftriaxone. The drugs are screened with a PAD designed to identify suspicious products, but this test must be backed up with confirmatory analysis by HPLC.6,15
- Arrival at Notre Dame: The in-country collaborators retain a portion of the pills in each package for their own analysis. The remaining pills are sent to the University of Notre Dame, Indiana, USA for intake where they are given unique identification codes, inspected, and prepared for distribution.
- Distribution: Prior to receiving samples for analysis, academic institutions must prove their ability to analyze a specific API. At ND, packages of pills are subdivided so some pills can be sent to participating DPAL locations for analysis; ND retains a portion of the pills for further testing.
- Analysis: Samples are analyzed via HPLC at DPAL locations, and the results are sent back to ND. Some samples are also analyzed in-house at ND or in the LMIC sites.
- Reporting: Results of analysis are sent back to ND and are compiled into a formal report that is sent to in-country stakeholders. When necessary, international SF medicines monitoring schemes including the WHO will be notified.
Data Generated from Workflow
At each step of the DPAL workflow, data are generated, and the existing metadata are updated. The data involved in each step are outlined below:
- Collection: During collection, each dosage form is assigned a temporary identification number and is attributed information on API, drug class, brand, manufacturer, country of origin, batch/lot number, API content, manufacturing date, and expiry date, as well as the overt or covert shopper’s name and town and location where the purchase was made. Historically, these data have been collected in a single spreadsheet of metadata. Pitfalls to this current arrangement include the manual entry of metadata creating typos, most often in lot/batch number, and the accumulation of multiple versions of the spreadsheets.
- Arrival at Notre Dame: An alert that samples are on their way to ND is sent from in-country to ND via email and the metadata spreadsheet is shared via Dropbox. Once the physical samples arrive at ND, they are assigned a new (and permanent) identification code, which is updated in the metadata spreadsheet and physically affixed to the sample. Challenges that can arise at this step include discovering missing or unaccounted for samples and recompiling shipping lists and lists of metadata for customs clearance.
- Distribution: Before receiving samples for analysis, participating DPAL locations must prove that their instrument is capable of accurately determining API concentration through a process called “system suitability,” which is a set of experiments based off of United States Pharmacopeia <1225> “Validation of compendial procedures” method that determines the instrument and method precision, accuracy, range, and other capabilities.16 Spreadsheet templates for system suitability are available to DPAL locations through OSF. These results are also shared back to the University of Notre Dame through OSF.
When a DPAL school is ready to analyze samples, they send an email to ND requesting a specific number of samples. Alternatively, ND will reach out to DPAL schools via email to see who has the capacity for more samples. From there, the ND DPAL coordinator will select dosage forms awaiting analysis and, in the case of dosage forms containing multiple pills, will split two pills from the dosage form and send those in a baggie to the requesting DPAL site. ND must retain a portion of the sample in case it requires further testing. The only sample information included in this shipment to DPAL schools is the unique identification code of the sample, API, and API content, which are physically affixed to the sample baggie. A shipping letter is included in the package, which lists unique identification codes of the samples as well as year and country of purchase of the samples. This shipping letter is historically the only record of the physical location of each sample. When a different DPAL school requests samples, a manual search of shipping letters is required to determine which samples are still at ND and awaiting analysis.
- 4. Analysis: Similar to system suitability, sample analysis spreadsheet templates are available on OSF for download and use by DPAL schools. Once analysis is completed, the finalized sample analysis spreadsheets, along with supplementary information including PDFs of the chromatographic results, are uploaded to OSF where they can be accessed by the DPAL coordinator. The results of these analyses and the spreadsheets themselves are then input into the existing metadata spreadsheet. This spreadsheet is only accessible to the DPAL manager. In-country stakeholders are not able to track the progress of a sample through analysis in real time, but must send requests for information about their samples via email or Dropbox. Unfortunately, this makes the system vulnerable, as errors can be introduced during data transfer from the sample analysis spreadsheet to the metadata spreadsheet, when results are manually input into the spreadsheets.
- 5. Reporting: The results of each sample’s analysis as compiled in the metadata spreadsheet are individually input into a manually typed report, which is shared with in-country stakeholders and regulatory agencies. A lack of automatic report generation or dashboard capabilities creates inefficiencies and workflows for sharing that do not scale well.
Current Limitations
It is crucial to be able to detect SF pharmaceuticals, as SF pharmaceuticals that go unnoticed can endanger health, prolong illness, cause mortality, promote drug resistance, and cause distrust in the medical system.1,2 While DPAL is not a certified pharmaceutical analysis laboratory and data produced cannot be used in a legal capacity to certify a medicine’s quality, the data generated through DPAL are used to report suspicious samples to regulatory agencies so they can conduct the compendial analysis that is needed to take action, such as recalling a product or banning a manufacturer from selling his or her products in the country. Results of the DPAL sample analysis are transmitted directly to stakeholders so they can make more informed decisions on purchasing certain brands and manufacturers.
As indicated above, DPAL data are created, managed, and accessed by multiple people in multiple locations with various editing permissions. A current limitation of the program is that the data generated through DPAL are stored in multiple types of file formats, including spreadsheet, PDF, and Word document, and are stored and shared on multiple different platforms, including OSF, Dropbox, email, personal computer, and external storage drive. In addition, changes are made to data by multiple users, and the existing methods of data management do not offer a way to track or log these changes. By combining all aspects of data management registered into a single blockchain ledger, this pilot project has potential to improve efficiency and throughput of sample analysis by streamlining data management into a single, secured platform.
Blockchain Technology
Blockchain is a distributed ledger shared across multiple locations that permanently records each datum entered with time and date stamps. It is attractive for its immutability, thus ensuring provenance and verifiability of data.13,14 While notably applied to cryptocurrencies and non-fungible tokens (NFTs), blockchain is also emerging as a solution for data management in medical applications.17–21 Blockchain technology has also been implemented to address challenges associated with the pharmaceutical supply chain, with a prominent goal being to combat SF medicines by tracking medicines as they traverse the global supply chain, allowing participants to identify the presence of SF products by greater visibility of rogue transactions.22–28
Blockchain is a good fit for managing DPAL data, as the DPAL process is distributed geographically throughout North America, Africa, and Europe. Blockchain offers solutions to DPAL’s current weaknesses, including the distribution of important data and communications across multiple file types and platforms, and a lack of data provenance when new information is added to the original metadata.
By introducing blockchain as a pilot project, we will have the benefit of testing the ledger with real samples and real users, recording feedback regularly to implement into the final form of the database before launching with all 30 schools and multiple in-county locations. The incorporation of blockchain provides a unique and flexible technology well-suited to the DPAL use case. Several significant fundamental advantages that the blockchain will contribute include the following:
- Creating a verifiable and immutable thread of all actions and data obtained associated with samples as they advance through the multi-step and multi-location process.
- Reliance on a trusted network of research institutions operating a ledger distributed among many countries and regions globally.
- Proof-of-authority method of confirming the legitimacy of all recorded data and activity, which operates with minimal overhead or energy consumption.
- Seamless interoperability with a workflow platform for managing data encryption and addressing on-chain and off-chain storage requirements.
- Ability to scale efficiently as sample volume and metadata generated escalate with the addition of laboratories and locations.
For the pilot, we selected three members of the current DPAL community which in microcosm together will allow us to test and confirm suitability of the system for handling samples from beginning-to-end. We chose to partner with ARTiFACTS to create this application to tap their experience and accelerate the timeline for confirming viability and moving forward with a broader deployment.
Integration of Blockchain into DPAL
The blockchain-based system designed by ARTiFACTS will be implemented in January 2022 with DPAL participants at Addis Ababa University, the University of Notre Dame, and Roanoke College. Regular feedback will be solicited from users and incorporated into further development of the ledger-based application for future implementation across all of DPAL.
Workflow
The workflow of a sample that is registered onto the blockchain ledger, beginning with sample creation at the time of purchase of the dosage form, and including workflows for sample shipment, analysis, and reporting of results, as well as the sample management dashboard, is outlined, as shown in Figure 2. The integration of blockchain into the DPAL process is described further in this section.
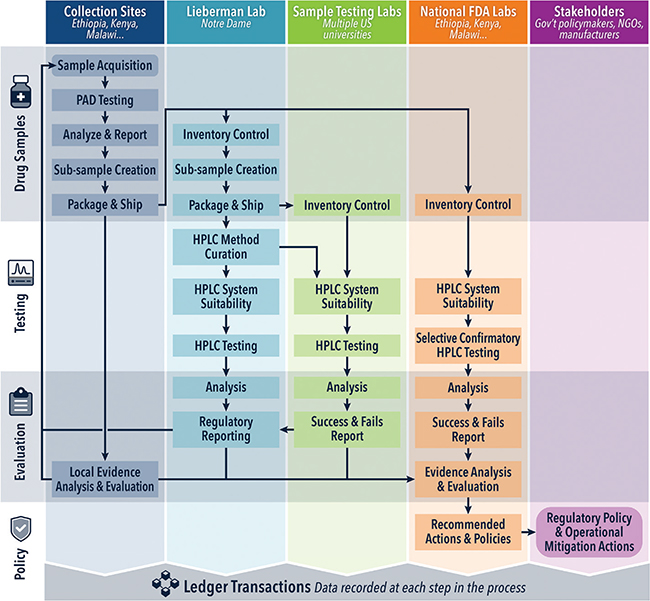
Fig. 2. Swim lane diagram detailing the sample analysis workflow through the blockchain ledger. HPLC: high performance liquid chromatography; PAD: paper analytical device.
Pharmaceutical samples are acquired at collection sites such as the lab in Addis Ababa, screened at the point of collection, and distributed to multiple testing laboratories. Sample inventory and stage in workflow are monitored by the ledger. Instrumental analysis methods such as HPLC are curated at the University of Notre Dame, and quality control measures such as systems suitability tests and pharmaceutical assay data and results are recorded at multiple lab sites, such as at Roanoke College. Data analysis and regulatory reporting are facilitated by cross-site information sharing. At each stage, the ledger records who has custody of each sample, what tests have been performed, and what the results were.
- Collection: After a pharmaceutical dosage form is purchased, a sample is created for it in the blockchain ledger. Once this sample is created, it will be assigned a unique identification code, and a label will be generated that can be affixed to the physical sample. In addition, all sample information that would have previously been manually input into the metadata spreadsheet for that sample will now be input directly into the blockchain database. This will include identification of the overt or covert shopper who purchased the sample. Typographical errors will be minimized using dropdown menu selections for all sample information, including brand, manufacturer, batch/lot number and more. In addition, the sample creator will be able to add new fields to each drop down menu – as there are thousands of possible drug manufacturers, brands, and batch/lot numbers that could be purchased. The sample and its information will be visible to the users: in-country lab manager, ND DPAL manager, and the relevant regulatory agencies. In addition, a record of when and by whom these changes were made will be recorded on the blockchain, creating an immutable history.
- Arrival at Notre Dame: Samples can be selected and grouped together as a package to be shipped at the University of Notre Dame. This establishes a record of the physical location of each sample. When the samples arrive at Notre Dame, they no longer need to be given new identifiers, while updates to blockchain record physical receipt – simplifying the intake process.
- Distribution: Now, requests for samples by DPAL schools will take place entirely on the blockchain-based system and recorded in the database, rather than through other channels like email, again streamlining where DPAL business takes place. Records of system suitability that prove a DPAL location’s ability to analyze a certain API will be housed and accessible to the DPAL manager on the blockchain. At ND, the samples that are being sent to a DPAL site will be split, both physically and on the blockchain, so that each sample has a new, unique identifier but will be traceable back to the original “parent” sample and its pertinent metadata. Again, multiple samples will be selected and grouped together as a package to be shipped to the DPAL site. Now, all information regarding the sample location, history, and metadata will be housed in the blockchain using a workflow application that streamlines the distribution of samples.
- Analysis: Results of analysis will be directly uploaded to the blockchain, including relevant attachments, including sample analysis spreadsheets and chromatographs of each sample. DPAL analysts will have permissions to upload their results, DPAL site coordinators, the DPAL manager, and in-country collaborators will be able to view where each sample is in its workflow as well as results of analysis in real time.
- Reporting: Results of analysis will be easily viewed by all relevant stakeholders on a dashboard of the blockchain database, including individual sample results and big picture information such as how many samples have been analyzed or are awaiting analysis, and trends in brand/manufacturer.
Users
As mentioned, there are many different users in DPAL who should have different rights to access and edit certain data within the DPAL data set. These users, their geographic location, responsibilities, and permissions on the blockchain ledger are described in Table 1.
Overall Benefits of the Blockchain Pilot
Blockchain offers clear benefits to DPAL data management compared with existing methods of data management. First, all data will be housed on a single platform, rather than being dispersed through Dropbox, OSF, email, external hard drive, and personal computer. This includes information about physical sample location, analysis results, and analysis procedural details. In addition, every piece of data will have date and time stamps, securing provenance of these data. This distributed ledger will be linked to an easy-to-use dashboard, so stakeholders will be able to view sample information in real time.
By combining all this information on a user-friendly platform, it will be easier to detect patterns of quality problems within and across different brands of products, so that regulators and purchasers can deal with problem brands proactively. This will also aid in developing enhanced statistical sampling techniques and best practices for routing work so that we can increase throughput, save cost for regulators, detect problem products faster, and impact fewer patients.
Limitations of the Blockchain Pilot
The pilot will connect the workflows of only one lab and one role within each lab. Potential operational challenges to overcome with the proposed pilot include (1) confirming skill requirements for users, (2) education and training of users, (3) achieving understanding and adoption of a ledger-based information system, (4) ability of personnel to capture and record all actions and metadata without disruption in a continuous chain of custody, (5) maintaining updated lists of persons authorized to access the ledger and enter data at different sites, and (6) challenges in the scalability and sustainability of the proposed system.
In addition, the pilot will retain a number of functional limitations, including the following:
- The pilot feature set will be small and will not be sufficient for broader deployment. A number of capabilities are deemed to be out of scope because they require more developmental efforts than can be completed in the time frame allotted to this project. Some of these include establishing interoperability with an Android mobile application created by DPAL for enhanced data capture, providing a protocol specification interface to expand the types of drugs tested and analyzed with this system, and blinding access to data and identities based on roles and clearance definitions.
- The pilot is expected to provide a valuable learning experience for both DPAL and ARTiFACTS that will surface new requirements and will guide the design and development of new functionality for broader deployment.
- The pilot design is scaled to support a limited number of users and locations. Broader deployment of this system will require the ability to operate at scale to support data capture from multiple nations and regional locations within different countries, multiple further testing laboratories required to perform confirmatory analyses, many users working simultaneously across multiple time zones, and a significant increase in the types of drugs processed and frequency/volume of drugs screened.
Conclusions
DPAL aims to aid in the detection of SF pharmaceutical products in LMICs using pharmaceutical analysis capabilities at academic institutions throughout the US and abroad. The data generated through DPAL regarding dosage form quality are used to inform in-country stakeholders when making purchasing options and in reporting suspicious samples to regulatory agencies for compendial analysis.
The use of blockchain technology for the management of DPAL data has the potential to increase the efficiency and throughput of DPAL, so that stakeholders can have results of post-market surveillance of pharmaceutical products in LMICs faster. The major intended benefits of using the blockchain include increasing efficiency by streamlining all data and communications into a single platform, securing provenance of data, and having an easy-to-use dashboard connected to the distributed ledger so stakeholders can view results of analysis in close to real time.
The initial pilot of this blockchain technology will include a subset of DPAL participants, including the DPAL manager at the University of Notre Dame (Indiana, USA), the in-country DPAL coordinator at Addis Ababa University (Addis Ababa, Ethiopia), and the DPAL site coordinator and DPAL analyst(s) at Roanoke College (Virginia, USA). Feedback from the pilot study will be regularly recorded and implemented into the widespread application of blockchain for all DPAL sites abroad and in the USA.
Contributors
Kathleen Hayes contributed to conceptualization, project administration, original draft preparation, reviewing, and editing of the article. Natalie Meyers and David Kochalko were involved in conceptualization, original draft preparation, and reviewing and editing of the manuscript. Christopher Sweet also contributed to conceptualization. Ayenew Ashenef and Tim Johann contributed to conceptualization, review and editing of the manuscript. Marya Lieberman was involved in conceptualization, supervision, original draft preparation, and review and editing of the manuscript.
Acknowledgments
The authors thank Kristina Davis for her contributions in designing Figure 2.
References
- World Health Organization. WHO Global Surveillance and Monitoring System for substandard and falsified medical products. Available from: https://apps.who.int/iris/bitstream/handle/10665/326708/9789241513425-eng.pdf?sequence=1&isAllowed=y [cited 13 December 2021].
- World Health Organization. A study on the public health and socioeconomic impact of substandard and falsified medical products. Available from: https://www.who.int/medicines/regulation/ssffc/publications/SEstudy-executive-summary-EN.pdf [cited 13 December 2021].
- Roth L, Bempong D, Babigumira JB, et al. Expanding global access to essential medicines: investment priorities for sustainably strengthening medical product regulatory systems. Global Health. 2018;14(1):1–2. https://doi.org/10.1186/s12992-018-0421-2
- Ndomondo-Sigonda M, Miot J, Naidoo S, Dodoo A, Kaale E. Medicines regulation in Africa: current state and opportunities. Pharmaceut Med. 2017;31(6):383–97. https://doi.org/10.1007/s40290-017-0210-x
- Bliese SL, Berta M, Lieberman M. Involving students in the distributed pharmaceutical analysis laboratory: a citizen-science project to evaluate global medicine quality. J Chem Educ. 2020 Oct 26;97(11):3976–83. https://doi.org/10.1021/acs.jchemed.0c00904
- Weaver AA, Reiser H, Barstis T, et al. Paper analytical devices for fast field screening of beta lactam antibiotics and antituberculosis pharmaceuticals. Anal Chem. 2013 Jul 2;85(13):6453–60. https://doi.org/10.1021/ac400989p
- Kochalko D, Morris C, Rollins J. Applying blockchain solutions to address research reproducibility and enable scientometric analysis. InSTI 2018 Conference Proceedings. Centre for Science and Technology Studies (CWTS); 2018 Sep 11, pp. 395–403. Available from: https://hdl.handle.net/1887/65349 [cited 22 February 2022].
- Heaven D. Bitcoin for the biological literature. Nature. 2019 Feb 1;566(7742):141–3. Available from: https://www.nature.com/articles/d41586-019-00447-9 [cited 22 February 2022].
- Kochalko D. Making the unconventional conventional: how blockchain contributes to reshaping scholarly communications. Inf Serv Use. 2019 Jan 1;39(3):199–204. https://doi.org/10.3233/ISU-190053
- Benchoufi M, Ravaud P. Blockchain technology for improving clinical research quality. Trials. 2017 Dec;18(1):1–5. https://doi.org/10.1186/s13063-017-2035-z
- Choudhury O, Sarker H, Rudolph N, et al. Enforcing human subject regulations using blockchain and smart contracts. Blockchain in Healthcare Today. 2018. Available from: https://blockchainhealthcaretoday.com/index.php/journal/article/view/10 [cited 15 December 2021].
- Javed IT, Alharbi F, Bellaj B, Margaria T, Crespi N, Qureshi KN. Health-ID: a blockchain-based decentralized identity management for remote healthcare. Healthcare. 2021;9(6):712–32. https://doi.org/10.3390/healthcare9060712
- Kuo T-T, Kim H-E, Ohno-Machado L. Blockchain distributed ledger technologies for biomedical and health care applications. J Am Med Inform Assoc. 2017 Nov 1;24(6):1211–20. https://doi.org/10.1093/jamia/ocx068.
- Attili S, Ladwa SK, Sharma U, Trenkle AF. Blockchain: the chain of trust and its potential to transform healthcare—our point of view. ONC/NIST Use of Blockchain for Healthcare and Research Workshop. Gaithersburg, MD: ONC/NIST; 2016. Available from: https://www.healthit.gov/sites/default/files/8-31-blockchain-ibm_ideation-challenge_aug8.pdf. [cited 22 February 2022].
- Bliese SL, Maina M, Were P, Lieberman M. Detection of degraded, adulterated, and falsified ceftriaxone using paper analytical devices. Anal Methods. 2019;11(37):4727–32. https://doi.org/10.1039/C9AY01489F
- United States Pharmacopeia. <1225> Validation of compendial procedures. 2021. Available from: https://latam-edu.usp.org/wp-content/uploads/2021/08/1225.pdf [cited 15 December 2021].
- Nakamoto, S. Bitcoin: a peer-to-peer electronic cash system. 2008; p. 9. Available from: https://bitcoin.org/bitcoin.pdf [cited 15 December 2021].
- Lennart A. Non-fungible token (NFT) markets on the Ethereum blockchain: temporal tevelopment, cointegration and interrelations (August 13, 2021). Available at SSRN: https://ssrn.com/abstract=3904683 or http://dx.doi.org/10.2139/ssrn.3904683
- Mettler M. Blockchain technology in healthcare: the revolution starts here. 2016 IEEE 18th International Conference on e-Health Networking, Applications and Services (Healthcom); 2016, pp. 1–3. https://doi.org/10.1109/HealthCom.2016.7749510
- Massaro M. Digital transformation in the healthcare sector through blockchain technology. Insights from academic research and business developments. Technovation. 2021;102386. https://doi.org/10.1016/j.technovation.2021.102386
- Mistry C, Thakker U, Gupta R, et al. MedBlock: an AI-enabled and blockchain-driven medical healthcare system for COVID-19. ICC 2021—IEEE international conference on communications; 2021, pp. 1–6. https://doi.org/10.1109/ICC42927.2021.9500397
- Taylor P. Applying blockchain technology to medicine traceability. Available from: https://www.securingindustry.com/pharmaceuticals/applying-blockchain-technology-to-medicine-traceability/s40/a2766/ -.WFnPQ7GZNzg [cited 22 February 2022].
- Mackey TK, Kuo TT, Gummadi B, et al. ‘Fit-for-purpose?’—challenges and opportunities for applications of blockchain technology in the future of healthcare. BMC Med. 2019 Dec;17(1):1–7. https://doi.org/10.1186/s12916-019-1296-7
- Sylim P, Liu F, Marcelo A, Fontelo P. Blockchain technology for detecting falsified and substandard drugs in distribution: pharmaceutical supply chain intervention. JMIR Res Protoc. 2018;7(9):e10163. https://doi.org/10.2196/10163
- Clauson K, Breeden EA, Davidson C, Mackey TK. Leveraging blockchain technology to enhance supply chain management in healthcare: an exploration of challenges and opportunities in the health supply chain. Blockchain Healthcare Today. 2018;1. https://doi.org/10.30953/bhty.v1.20
- Tseng JH, Liao YC, Chong B, Liao SW. Governance on the drug supply chain via gcoin blockchain. Int J Environ Res Public Health. 2018;15(6):1055. https://doi.org/10.3390/ijerph15061055
- Vruddhula S. Application of on-dose identification and blockchain to prevent drug counterfeiting. Pathog Glob Health. 2018;112(4):161. https://doi.org/10.1080/20477724.2018.1503268
- Mackey TK, Nayyar G. A review of existing and emerging digital technologies to combat the global trade in fake medicines. Expert Opin Drug Saf. 2017;16(5):587–602. https://doi.org/10.1080/14740338.2017.1313227
Copyright Ownership: This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, adapt, enhance this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0.